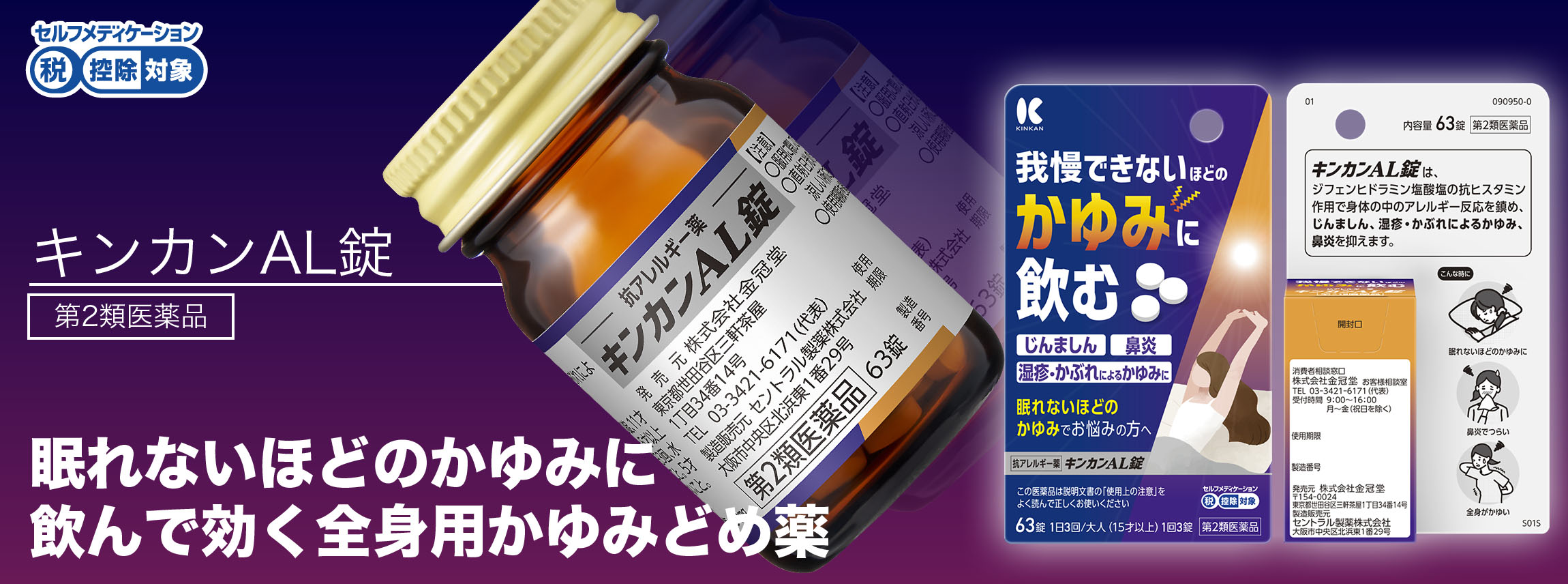
KINKAN AL Tablet

KINKAN AL Tablet
Second-class OTC drugs
Form: Tablet
Capacity: 63 tablets
Features
KINKAN AL tablets are an oral allergy medication that can be used for overall itching. Diphenhydramine hydrochloride, which has an antihistamine effect, soothes allergic reactions throughout the body and suppresses symptoms such as hives, rhinitis, or itching caused by eczema/rash.
Effect/efficacy
Hives, itching due to eczema/rash, rhinitis
Dose and administration
Take the following dose three times a day with water or warm water.
| Age | Single dose |
|---|---|
| Adults (15 years and above) | 3 tablets |
| 11 years old or older and under 15 years old | 2 tablets |
| 5 years old or older and under 11 years old | 1 tablet |
| Children under 5 years old | Do not take |
● It can be taken any time, before or after meals.
(It is recommended to take the medicine at least 4 hours apart)
<Notes on dose and administration>
- (1) Please strictly follow the directions and dosage of the medicine.
- (2) If you are giving this medicine to children, have them take it under the supervision of a parent or guardian.
Ingredient and amount
| Ingredient | Capacity [per 9 tablets] |
|---|---|
| Diphenhydramine hydrochloride | 90 mg |
Contains cellulose, lactose, carmellose calcium, magnesium stearate, hypromellose, macrogol, titanium oxide, and carnauba wax as an additives.
Caution
* For notes on dose and administration, refer to the dose and administration section.
■Caution for use■
[Don’t] (If instructions/directions are not followed, conditions may worsen or it is easier to get adverse effects )
- 1. Do not use any of the following medicines while taking this drug.
Other allergy medications, oral medications containing antihistamines (cold medicines, cough suppressants and expectorants, oral medications for rhinitis, motion sickness medication, etc.) - 2. Do not drive or operate machinery after taking this medicine. (Drowsiness may occur).
- 3. If you are breastfeeding, do not take this medicine or avoid breastfeeding if you take this medicine.
- 4. Do not drink alcohol before or after taking this medicine.
- 5. Do not take this medicine for a long time.
[Consult]
- 1. The following persons should consult a physician, pharmacist or registered sales clerk before using this product.
- (1) Those who are under treatment by a physician.
- (2) Those who are pregnant or might be pregnant.
- (3) Those who have ever had drug induced allergic reactions.
- (4) Those who have following symptom
Difficulty in urination - (5) Those who have been diagnosed with
Glaucoma
- 2. If the following adverse effects occur after taking this medicine, stop taking it immediately, and consult a physician or a pharmacist or registered sales clerk with this package.
Area concerned Symptoms Skin Eruption, redness, itching Digestive organ Nausea, vomiting, loss of appetite Urinary organ Difficulty in urination - 3. When the following symptoms occur after taking this medicine and if any of these symptoms persist or worsen, stop taking this medicine and consult a physician or a pharmacist or registered sales clerk with this package.
Dry mouth, drowsiness - 4. If symptoms do not improve after taking this medicine for 5-6 days, stop taking it and consult a physician, pharmacist or registered sales clerk with this package.
■Notes on storage and handling■
- (1) Store out of the reach of children.
- (2) Tightly close the cap and store it in a cool dry place out of direct sunlight.
- (3) Do not replace it with another container. (It may cause misuse or affect the product quality.)
- (4) If tablets contain moisture, do not drop water on the product or touch it with wet hands, as this may cause the ingredients to change. If the tablets get wet by mistake, discard the wet tablets.
- (5) If the bottle cap is not closed properly, moisture may affect the quality of the medicine, so please make sure to close the bottle cap tightly after each dose.
- (6) The plastic packaging inside the bottle is there to protect the tablets from damage during transport. Please discard it after opening.
- (7) Do not take the product after the expiration date.